Lewis Structure and Discussion on Open-Chain and Cyclical D-Glucose
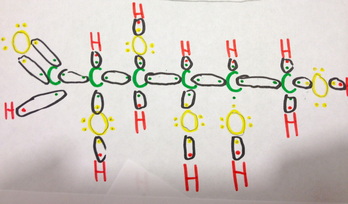
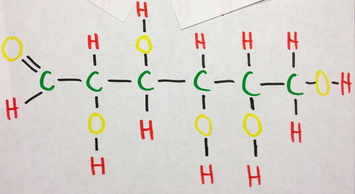
D-Glucose occurs in multiple forms in nature, one of which being the open chain structure shown here. The first picture is the process of making a Lewis structure, by first diagramming the valence electrons of all the atoms in the molecule. The black circles imply that these electrons will bond, and these conds are shown in the latter picture. Almost all of the bonds are single bonds, meaning the atoms are sharing one electron from each other but, one bond between Carbon and Hydrogen on the end of the carbon chain is a double bond. Next, it is possible to delve into the nature of these bonds. The bonds between carbon and carbon are completely covalent as the electronegativity's difference is 0. On the other hand, the bond between carbon and hydrogen is strongly covalent with the difference being only 0.3. The bonds between carbon and oxygen are moderately covalent, with the difference being 1.0. This bond is also polar. Lastly, the bond between oxygen and hydrogen is moderately to weakly covalent, as the difference is 1.3. This bond is also polar. Since this molecule occurs in a chain, the molecular geometry is linear, and of 180 degrees without a defined VSEPR form.
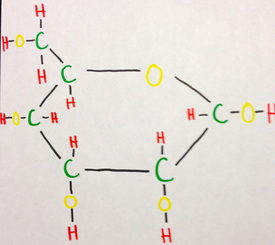
Cyclical or a-D-Glucopyranose
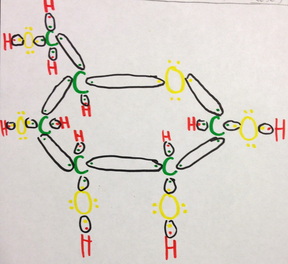
Glucose molecules often have carbon-based rings, like in the form of a-D-Glucopyranose. It has a hexagonal shaped ring consisting of 5 carbon atoms and one oxygen with hydroxide and a hydrocymethal group of CH2OH. The same kinds of bonds occur as in the open-chain model, however without any double bonds. The bond of carbon to carbon is completely covalent, the bond of carbon to hydrogen is strongly covalent, the bond of carbon to oxygen is polar moderately covalent, and the bond of oxygen to hydrogen is polar weakly covalent.
Color Key for Above Diagrams